Buy Fitaro online – Generic Semaglutide at a Reasonable Price
| Product Name: | Fitaro (Semaglutide) injection |
|---|---|
| Dosage: | 0.25 mg, 0.5 mg, 1 mg, 1.7 mg, 2.4 mg |
| Best Price: | $60.00 |
| Product Name: | Rybelsus (Semaglutide oral) |
|---|---|
| Dosage: | 3 mg, 7 mg, 14 mg |
| Best Price: | $199.00 |
| Where To Buy Online | Order Online |
Contents:
- What You Need to Know About Fitaro?
- How does Fitaro Work?
- Main Indications for Using Fitaro
- Who Can Benefit from Fitaro?
- Is Fitaro a Real Alternative to Ozempic and Wegovy?
- How to Administer Fitaro
- How Long Does It Take to See Results?
- Risks and Side Effects of Fitaro
- What Should Not Be Combined with Fitaro
- Price and Availability of Fitaro
- Do You Need a Prescription to Get It?
- Generic Alternatives and Their Availability
- What to Know About Long-Term Use of Fitaro
What You Need to Know About Fitaro?
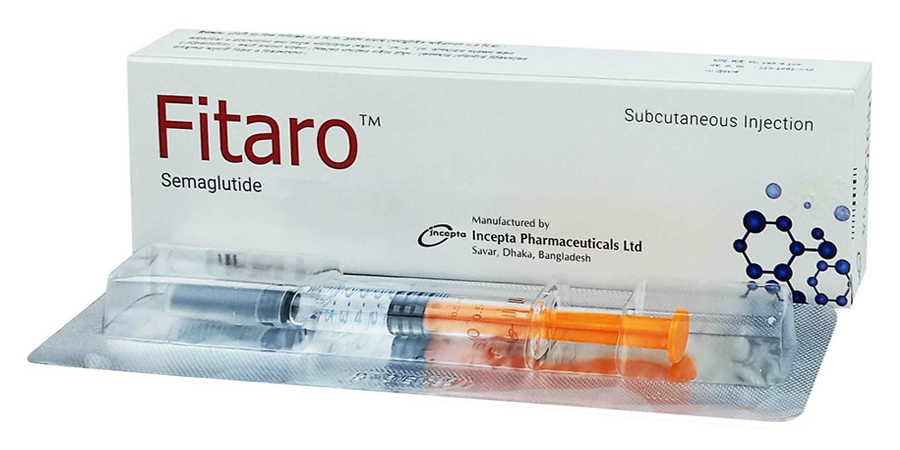
Fitaro is a trade name for an injectable medication based on semaglutide, a member of the glucagon-like peptide-1 (GLP-1) receptor agonist class. It is used to treat type 2 diabetes and to support weight management in adult patients with obesity or overweight accompanied by metabolic disorders. Semaglutide is an analog of human GLP-1 that is resistant to degradation by the DPP-4 enzyme, providing prolonged action and allowing for once-weekly administration.
The pharmacological activity of Fitaro is based on its ability to activate GLP-1 receptors, which leads to stimulation of insulin secretion in response to food intake, suppression of glucagon release, appetite reduction, and delayed gastric emptying. As a result, patients benefit from both lower blood glucose levels and moderate but sustained weight loss. This makes Fitaro particularly relevant for type 2 diabetic patients with obesity—those at highest risk for cardiovascular complications and metabolic disturbances.
Fitaro is manufactured by Incepta Pharmaceuticals Ltd. and comes in prefilled pen injectors for subcutaneous use. Available dosages include 0.25 mg, 0.5 mg, 1 mg, 1.7 mg, and 2.4 mg. Treatment typically begins with a low dose of 0.25 mg weekly, followed by gradual dose escalation over several weeks to reach therapeutic effectiveness with minimal risk of side effects.
Although initially used exclusively for diabetes, since 2021 semaglutide has been actively incorporated into weight management protocols. Its clinically proven effectiveness in weight reduction, along with positive effects on fasting glucose and HbA1c, makes Fitaro a dual-action drug capable of replacing several medications in patients with comorbid conditions.
It is important to understand that Fitaro is not a "quick fix" and is not intended for emergency glucose or weight reduction. Its effects develop gradually, in combination with dietary recommendations and physical activity. The average time to noticeable clinical improvement is between 4 and 8 weeks after therapy initiation, with optimal results achieved through consistent lifestyle control and adjunctive treatment for metabolic disorders.
In summary, Fitaro is a modern, effective, and thoughtfully designed medication aimed at comprehensive management of diabetes and obesity. For many, this is also a convenient way to buy generic Ozempic — a well-known medication based on semaglutide, whose efficacy is supported by international clinical guidelines. Considering that Fitaro contains the same active ingredient, semaglutide, its use can be seen as a rational choice to buy generic semaglutide in a similarly reliable form at a more affordable price. It opens new therapeutic possibilities for patients who previously failed to achieve stable glucose control and weight loss through standard approaches.
How Does Fitaro Work?
The pharmacological action of Fitaro is based on the active ingredient semaglutide, which mimics the natural hormone GLP-1 (glucagon-like peptide-1) secreted by the gut in response to food intake. GLP-1 plays a key role in glucose regulation, insulin secretion, appetite control, and feeding behavior. In the human body, this hormone is rapidly degraded by the enzyme dipeptidyl peptidase-4, limiting its natural effect. Semaglutide in Fitaro is resistant to this degradation, providing sustained action with just one weekly injection.
After subcutaneous administration, Fitaro enters the systemic circulation and binds to GLP-1 receptors located on pancreatic, gastric, brain, cardiovascular, and other cells. This initiates a cascade of biochemical processes aimed at metabolic normalization.
The primary effect is stimulation of insulin secretion by pancreatic β-cells. Unlike insulin therapy, Fitaro induces insulin release only in the presence of blood glucose, thus reducing the risk of hypoglycemia. Another key mechanism is suppression of glucagon secretion, which decreases glucose release from liver stores and lowers fasting blood sugar and post-meal glucose spikes.
Fitaro also slows gastric emptying, reducing carbohydrate absorption rates and smoothing postprandial glycemic curves. This prevents sharp glucose fluctuations, which is especially important in insulin-resistant patients.
Beyond glycemic control, Fitaro affects appetite-regulation centers in the hypothalamus, suppressing hunger and reducing compulsive eating. Patients frequently report spontaneous reductions in food intake and decreased cravings for high-calorie foods. This results in lower caloric consumption and gradual weight loss.
Fitaro also has cardiometabolic benefits. It can lower blood pressure and triglyceride levels, improving lipid profiles. This is particularly important for patients with metabolic syndrome, where diabetes is accompanied by hypertension, obesity, and atherosclerosis. These effects arise from both direct receptor action and the secondary benefits of weight loss and improved insulin sensitivity.
Thus, Fitaro delivers comprehensive metabolic effects: it stabilizes glycemia, regulates appetite, promotes weight loss, improves lipid profiles, and reduces vascular risk. All effects are interconnected and develop sequentially, making it one of the most versatile and clinically valuable therapies for modern diabetes and obesity treatment.
Main Indications for Using Fitaro
Fitaro is indicated for adult patients with confirmed type 2 diabetes to improve glycemic control in addition to diet and physical activity. This is its primary approved use according to regulatory agencies. It may be used as monotherapy in patients intolerant to other antidiabetic drugs, or in combination with metformin, sulfonylureas, SGLT-2 inhibitors, and even insulin.
However, the therapeutic potential of Fitaro goes beyond glycemic control. A second major indication is long-term weight management. The drug can be prescribed to adults with a body mass index (BMI) ≥30 kg/m² (obesity) or ≥27 kg/m² (overweight) in the presence of at least one associated condition such as hypertension, dyslipidemia, obstructive sleep apnea, or prediabetes. In such cases, Fitaro is used for long-term pharmacologic weight reduction when diet and exercise alone are insufficient.
Fitaro is not intended for type 1 diabetes or diabetic ketoacidosis. It is not approved for pediatric use, and it is contraindicated in patients with severe gastrointestinal disorders, a history of pancreatitis, or C-cell thyroid tumors.
Regardless of the indication, Fitaro is prescribed as part of a comprehensive therapy plan. It does not replace a healthy lifestyle but enhances it by providing physiological support for blood sugar and appetite regulation. Its multifaceted effects and evidence-backed outcomes make it a preferred choice in modern treatment strategies for diabetes and obesity.
Who Can Benefit from Fitaro?
Fitaro is intended for patients whose metabolic issues are not adequately managed by standard methods. Primarily, this includes individuals with type 2 diabetes who continue to experience elevated glucose levels despite diet, physical activity, and oral hypoglycemic therapy. It is also suitable for patients struggling with appetite control and body weight, even when adhering to all medical recommendations.
The following groups benefit most from Fitaro:
- Individuals with type 2 diabetes needing to enhance the effect of metformin or replace medications associated with a high risk of hypoglycemia.
- Patients with obesity (BMI ≥30), especially those with comorbidities such as hypertension, fatty liver disease, or obstructive sleep apnea.
- People with food addiction or impaired satiety response.
- Patients with prediabetes who can delay or prevent diabetes through weight loss.
- Individuals at high cardiovascular risk—studies show semaglutide reduces cardiovascular events in this population.
Fitaro is especially useful for those with severe insulin resistance, nighttime hunger, nocturnal hyperglycemia, or compulsive overeating. By acting on central nervous system receptors, it suppresses abnormal food cravings and helps patients establish new eating patterns without relying on constant willpower.
Fitaro may also be considered as a non-invasive alternative to bariatric surgery in early-stage candidates who are hesitant to undergo surgical procedures. When dosed correctly and combined with lifestyle interventions, weight loss achieved with Fitaro may be comparable to that of surgery—without the risks or extended recovery periods.
In summary, Fitaro is a treatment option for patients with pronounced metabolic decompensation who need to regain control over glucose levels, appetite, and weight without resorting to aggressive therapeutic measures.
Is Fitaro a Real Alternative to Ozempic and Wegovy?
Fitaro is positioned as a full-fledged alternative to both Ozempic® and Wegovy®, as it contains the same active ingredient—semaglutide. It addresses both therapeutic goals: glycemic control in type 2 diabetes and weight loss in patients with obesity. This makes Fitaro one of the few medications capable of replacing two original Novo Nordisk brands, particularly in regions with limited access to branded drugs.
Pharmacological Equivalence
Fitaro’s active substance—semaglutide—is identical to that used in Ozempic and Wegovy. It has high receptor selectivity for GLP-1 and resistance to enzymatic degradation by DPP-4. Its pharmacokinetics, weekly dosing regimen, and available strengths are fully comparable. Fitaro is available in doses up to 2.4 mg, which aligns with Wegovy’s indication for weight management. This allows Fitaro to be used both in diabetology and obesity treatment—unlike Ozempic, which is officially registered only for diabetes.
Clinical Effectiveness
Since semaglutide is chemically identical regardless of brand, Fitaro’s effectiveness, when properly administered, matches that of the original products. Clinical data confirm that Fitaro reduces HbA1c, body weight, blood pressure, and improves lipid profiles. This means that, assuming proper manufacturing and storage conditions, Fitaro can deliver the same therapeutic outcomes as the more expensive branded drugs.
Financial and Logistical Alternative
Fitaro stands out for its affordability—often costing 2 to 4 times less than the originals. It is available in Asia, Latin America, Africa, and the Middle East, where Ozempic and Wegovy are often inaccessible due to price or supply issues. This makes Fitaro a realistic option for patients requiring long-term semaglutide therapy who cannot afford or obtain branded products.
Fitaro is a true alternative to Ozempic and Wegovy in terms of composition, indications, and efficacy. It is especially valuable in regions where branded drugs are unavailable or unaffordable. With proper use and monitoring, Fitaro can achieve equivalent clinical results.
How to Administer Fitaro
Proper administration of Fitaro is critical for achieving therapeutic effectiveness and minimizing side effects. The drug is injected subcutaneously once a week, regardless of food intake or time of day. It is recommended to select a specific day of the week and consistently adhere to it—this helps maintain stable blood levels and simplifies adherence to the treatment regimen.
Treatment with Fitaro always starts with a minimal dose of 0.25 mg once weekly. This dose has limited therapeutic effect but allows the body to adapt and reduces the likelihood of gastrointestinal side effects. After four weeks, the dose is increased to 0.5 mg weekly. If tolerance is good and glycemic targets are not met, the dose may be increased to 1 mg. Further titration to 1.7 mg and 2.4 mg per week is possible—these are primarily used in weight management protocols.
The injection is administered subcutaneously into the abdomen, upper arm, or front thigh. Intramuscular or intravenous administration is not recommended. The injection site should be rotated to avoid irritation and lipodystrophy.
If a dose is missed, it should be taken as soon as possible within 5 days. If more than 5 days have passed, the dose should be skipped, and the regular schedule resumed. Doubling the dose to compensate is not allowed.
Administration notes:
- Do not massage the injection site after administration.
- The medication should be at room temperature before use.
- The pen should not be frozen, overheated, or shaken.
Fitaro can be used as monotherapy or in combination with other agents—such as metformin, SGLT-2 inhibitors, or basal insulin. In such cases, regular glucose monitoring is required to avoid hypoglycemia, especially at treatment initiation.
There is no time limit for Fitaro treatment—it is used as long as clinical benefit is maintained. Discontinuation is possible but may lead to symptom recurrence and worsened glycemic or weight control.
Patients should be trained in proper injection technique and receive clear medical guidance on dose escalation. Self-adjustment of dose or schedule is strictly prohibited. In cases of nausea, vomiting, or appetite suppression after a dose increase, it is permissible to temporarily revert to the previous dose—only after consulting the prescribing physician.
How Long Does It Take to See Results?
Fitaro is a medication with gradual but sustained effects. Its pharmacokinetics involve a slow buildup of plasma concentration and a delayed onset of clinical benefits. This must be considered when evaluating therapy outcomes—immediate reductions in glucose or body weight should not be expected.
Glycemic improvement, such as reduction in fasting and postprandial blood glucose, typically begins within the first 4 weeks of treatment upon reaching a dose of 0.5 mg. However, maximum glycemic effect is observed at doses of 1 mg or higher, usually by week 8–12 of therapy. Clinical trials confirm that by this time, most patients experience a significant drop in HbA1c and achieve stable glucose levels within target ranges.
As for weight loss, initial changes may become noticeable by weeks 6–8. Weight reduction occurs progressively, without sharp drops or plateaus, provided that dietary recommendations are followed. On a 2.4 mg dose, average weight loss over 6 months reaches 10–15% of baseline, though individual outcomes may vary.
It is important to understand that Fitaro has a cumulative effect: the longer the patient remains on therapy, the more pronounced the clinical results. Peak therapeutic effect generally occurs around months 5–6, and in some cases, later—especially in patients with obesity where metabolic remodeling requires more time.
Factors that influence the speed of response include:
- baseline glucose level and body weight;
- comorbidities (e.g., insulin resistance, hypothyroidism);
- individual metabolism and receptor sensitivity;
- adherence to diet and physical activity;
- strict compliance with dose escalation schedule.
Effectiveness is typically assessed at 3 and 6 months based on a range of parameters: fasting glucose, HbA1c, body weight, BMI, waist circumference, and lipid profile. If no progress is observed, the physician may adjust the dose, add medications, or revise the nutrition plan.
In summary, Fitaro is not a fast-acting drug but a tool for systemic metabolic correction. Its effects appear over 1 to 6 months and are maintained with long-term use. Patience, consistency, and adherence to the treatment plan are key to achieving lasting results.
Risks and Side Effects of Fitaro
Despite its proven effectiveness, Fitaro may be associated with side effects. Most of them result from the drug's mechanism of action—its effects on the gastrointestinal tract, appetite regulation centers, and metabolism. In the vast majority of cases, adverse effects are transient, arise during dose escalation, and resolve as the body adapts.
The most common side effects include:
- Nausea – occurs in over 30% of patients, especially during the first few weeks or when increasing the dose. It may worsen with rapid eating, overeating, or high-fat meals.
- Vomiting – less frequent, mostly in sensitive individuals. May require temporary dose reduction or returning to a previous dose level.
- Diarrhea or constipation – related to slowed gastrointestinal motility. May require dietary adjustments or symptomatic treatment.
- Appetite suppression – a physiological effect often considered beneficial, but in some patients it may lead to inadequate caloric intake and nutrient deficiency.
Less common side effects:
- hypoglycemia – mostly when combined with insulin or sulfonylureas;
- headache, fatigue, weakness;
- elevated amylase and lipase levels;
- injection site irritation;
- dehydration due to severe nausea or vomiting.
Serious and rare risks include:
- acute pancreatitis – a rare complication requiring immediate drug discontinuation;
- C-cell thyroid tumors (medullary thyroid carcinoma) – observed only in rodent studies but contraindicated in individuals with a family history of such tumors;
- gallstones – may occur with rapid weight loss;
- renal impairment secondary to severe dehydration.
Patients should be informed of warning signs such as sudden abdominal pain, persistent vomiting, jaundice, or urinary disturbances. These require immediate medical evaluation and possible discontinuation of therapy.
To minimize side effects, it is essential to strictly follow the stepwise titration schedule and avoid unauthorized changes in dosing or intervals. The physician must monitor tolerability and adjust the regimen if needed.
What Should Not Be Combined with Fitaro
Despite its receptor selectivity, Fitaro can interact with certain medications, especially in the context of combination therapy for diabetes or obesity. The primary mechanism of interaction involves delayed gastric emptying, which may affect the absorption and efficacy of other drugs.
Drug categories not recommended for use with Fitaro:
- Oral drugs with a narrow therapeutic index: such as warfarin, digoxin, and lithium. Slower absorption may lead to fluctuating concentrations and unstable therapeutic effects.
- Antibiotics and antiretroviral agents: their efficacy may be reduced due to impaired absorption.
- Insulins and sulfonylureas: when combined with Fitaro, the risk of hypoglycemia increases. Dose adjustments and frequent glucose monitoring are required.
- Other GLP-1 receptor agonists: simultaneous use is strictly contraindicated due to the risk of cumulative overdose and severe adverse effects.
- Gastrointestinal irritants: NSAIDs, corticosteroids, potassium tablets may increase the risk of gastritis or mucosal erosion.
In addition to medications, interactions with food and alcohol should be considered:
- Alcohol may exacerbate gastrointestinal side effects and increase the risk of hypoglycemia. Alcohol consumption during Fitaro therapy is discouraged.
- Fatty, spicy, and large meals raise the likelihood of nausea, particularly during early treatment. A fractional, moderate diet is recommended.
Fitaro may also affect the absorption of oral hormonal contraceptives—during the first 4 weeks of treatment, their effectiveness may decrease. Barrier methods are recommended during this period.
Before starting Fitaro, all current medications—including OTC drugs and supplements—must be disclosed to the healthcare provider. Adjustments to administration timing or regimen may be required to avoid complications and ensure safe therapy.
Price and Availability of Fitaro
The price of Fitaro depends on the dosage, country of sale, form of packaging, and the pricing policy of the pharmacy or online platform. Since the drug is available in prefilled pens with fixed doses of active ingredient, the cost increases proportionally with higher concentrations. At the time of writing, the approximate price per Fitaro pen ranges from $90 to $250, depending on dosage strength and region.
In most countries, the drug is sold by prescription through pharmacy chains, including specialized diabetes centers and hormone distributors. Some online pharmacies offer Fitaro delivery, but in such cases, it is essential to verify the site’s legality, storage conditions, and quality certifications.
Fitaro's availability varies significantly. In the United States and the EU, it is widely accessible under insurance plans and private obesity treatment programs. In developing countries, availability may be limited, and prices higher due to lack of insurance coverage and import duties. In regions with less strict pharmaceutical oversight, Fitaro may be sold by private suppliers without authenticity guarantees.
In some pharmacies, Fitaro is included in reimbursement lists under diabetes support programs. Certain countries also provide public subsidies for GLP-1 class medications, allowing patients to cover only part of the cost.
Additional costs may include:
- supplies (needles, alcohol wipes, disposal containers);
- laboratory monitoring (HbA1c, blood chemistry);
- endocrinologist consultations;
- supportive medications for side effects (antiemetics, antispasmodics, etc.).
Thus, the total cost of Fitaro therapy consists not only of the drug price but also of various accompanying expenses. However, considering the reduced risk of cardiovascular events, weight loss benefits, and potential insulin discontinuation, the overall economic value remains high even with a substantial initial investment.
Do You Need a Prescription to Get It?
Fitaro is a prescription-only medication. In all regulated pharmaceutical markets—including the U.S., Canada, the U.K., and EU countries—it is illegal to sell Fitaro without a doctor's prescription. This requirement stems from the drug’s mechanism of action, risk of serious side effects, and the need for dose titration under medical supervision.
The prescription status also accounts for potential interactions with other antidiabetic drugs, the need for individual tolerability assessment, and contraindications such as:
- history of pancreatitis or thyroid tumors;
- severe gastrointestinal conditions (e.g., gastroparesis);
- hepatic or renal insufficiency;
- allergy or hypersensitivity to any ingredient in the drug.
A prescription for Fitaro can be issued after a consultation with an endocrinologist or general practitioner. Typically, the following are required:
- confirmed diagnosis of type 2 diabetes or obesity (BMI ≥27);
- medical history of previous treatments;
- lab results (glucose, HbA1c, lipids, creatinine, ALT/AST);
- risk evaluation, including cardiovascular and oncological profiles.
In some countries, electronic prescriptions are accepted at both retail and online licensed pharmacies. However, attempts to purchase Fitaro without a prescription—especially from unverified websites—pose risks of receiving counterfeit products, incorrect dosing, and lack of clinical oversight.
It is also critical to understand that self-medicating with Fitaro is unacceptable. Without proper medical supervision, there is a high risk of hypoglycemia, dehydration, nutritional deficiencies, gastrointestinal complications, and unpredictable drug interactions. Only a specialist can accurately determine the appropriate dose, injection frequency, and compatibility with other medications.
In summary, Fitaro requires mandatory medical supervision. A prescription is not just a formality but a vital safety measure ensuring effective and controlled therapy.
Generic Alternatives and Their Availability
Fitaro is a generic version of Ozempic®, the original semaglutide produced by Novo Nordisk. Both products contain the same active substance—semaglutide—and share identical therapeutic indications, mechanisms of action, and administration schedules. The main differences lie in the manufacturer and price: Fitaro is produced by Incepta Pharmaceuticals (Bangladesh) and targeted at markets where Ozempic’s patent is either expired or not strictly enforced, such as outside the U.S. and EU.
Fitaro was introduced as a licensed or replicated version of Ozempic, designed for developing countries where many patients cannot afford high-cost original brands. This is standard practice in global pharmaceuticals—generics are widely distributed in regions without patent barriers, providing access to effective molecules for larger populations.
Comparison overview:
- Ozempic® – the original semaglutide by Novo Nordisk. Manufactured in Denmark and the U.S., registered by the FDA, EMA, Health Canada, MHRA, and other major regulatory bodies.
- Fitaro – generic semaglutide by Incepta Pharmaceuticals. Supplied in pen injectors with 0.25 mg, 0.5 mg, 1 mg, 1.7 mg, and 2.4 mg doses, covering both diabetic and anti-obesity therapies.
Fitaro is used in clinical practice and shows equivalent effectiveness when the treatment protocol is followed.
Other potential semaglutide generics are under development. However, full replacement of Ozempic in countries with active patent protection is not yet possible. Only after expiration of those patents (approximately after 2031) can a broad market of generics be expected.
It’s also important to distinguish among semaglutide formulations:
- Ozempic® and Fitaro – injectable forms used for diabetes and weight management.
- Rybelsus® – oral semaglutide from Novo Nordisk, currently without generics.
- Wegovy® – semaglutide 2.4 mg, officially registered for obesity treatment as a high-dose variant.
Fitaro remains the only available generic semaglutide in some countries, offering cost-effective access to proven therapy. When choosing between a branded and generic version, it’s essential to consider the registration country, manufacturing license, pharmacovigilance, and medical guidance.
What to Know About Long-Term Use of Fitaro
Long-term use of Fitaro is not only acceptable but often necessary as part of therapeutic strategy. Type 2 diabetes and obesity are chronic conditions that cannot be cured but require continuous management. Therefore, Fitaro therapy may last for months or even years without interruption.
Clinical trials and post-marketing studies show that Fitaro maintains stable efficacy over extended use, continuing to improve metabolic parameters. Reductions in glucose, HbA1c, body weight, blood pressure, and lipid levels are confirmed even after 1–2 years of continuous therapy.
Benefits of long-term use include:
- stable control of blood sugar and weight without fluctuations;
- reduced cardiovascular risk in diabetic patients;
- lower need for insulin or additional medications;
- improved quality of life, including eating behavior and sleep;
- prevention of prediabetes progression to diabetes.
However, prolonged use requires adherence to the following:
- regular medical monitoring (glycemia, biochemistry, weight control);
- tolerability assessments—especially gastrointestinal, liver, and kidney function;
- dietary and physical activity support;
- prevention of deficiencies (possible shortfalls in vitamins, protein, iron due to reduced appetite);
- periodic review of the treatment plan and dose adjustments by a physician.
Some patients may report a perceived loss of effect during long-term therapy. This is usually due to dietary lapses, decreased activity, or psychological fatigue—not drug tolerance. The solution is adjusting the treatment strategy, not discontinuing the medication.
Fitaro does not cause physical dependence, but stopping treatment without alternatives often leads to weight regain and glycemic deterioration. Therefore, discontinuation should only occur for medical reasons and under a transition plan to another treatment approach.
In conclusion, Fitaro is suitable for long-term, continuous use under appropriate medical supervision. It offers systemic, multi-level benefits in diabetes and obesity management, minimizes disease progression, and provides patients with long-term therapeutic prospects.

 Call Us
Call Us